Lewis Dot Diagram for Nitrogen: Step-by-Step Instructions
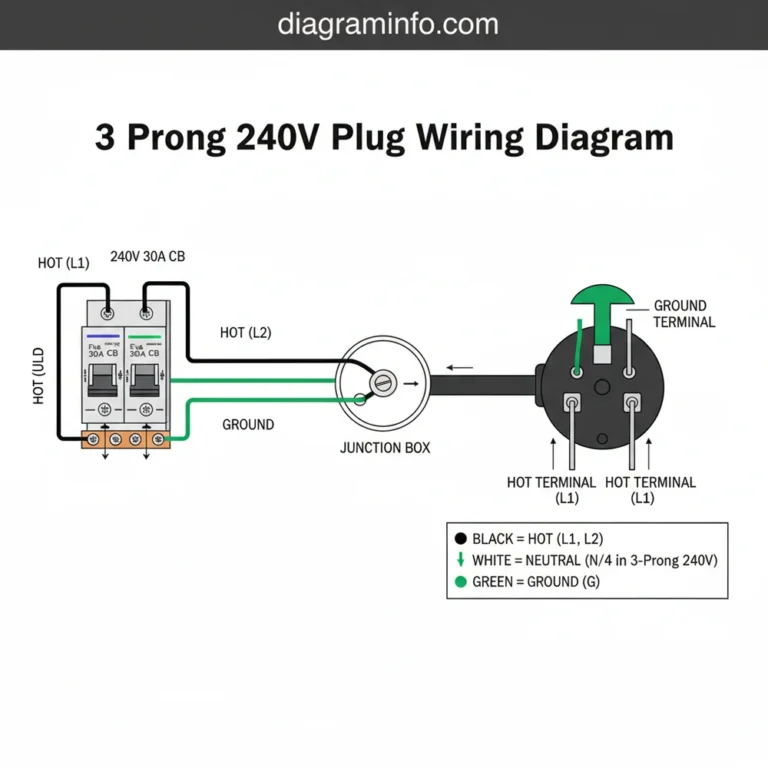
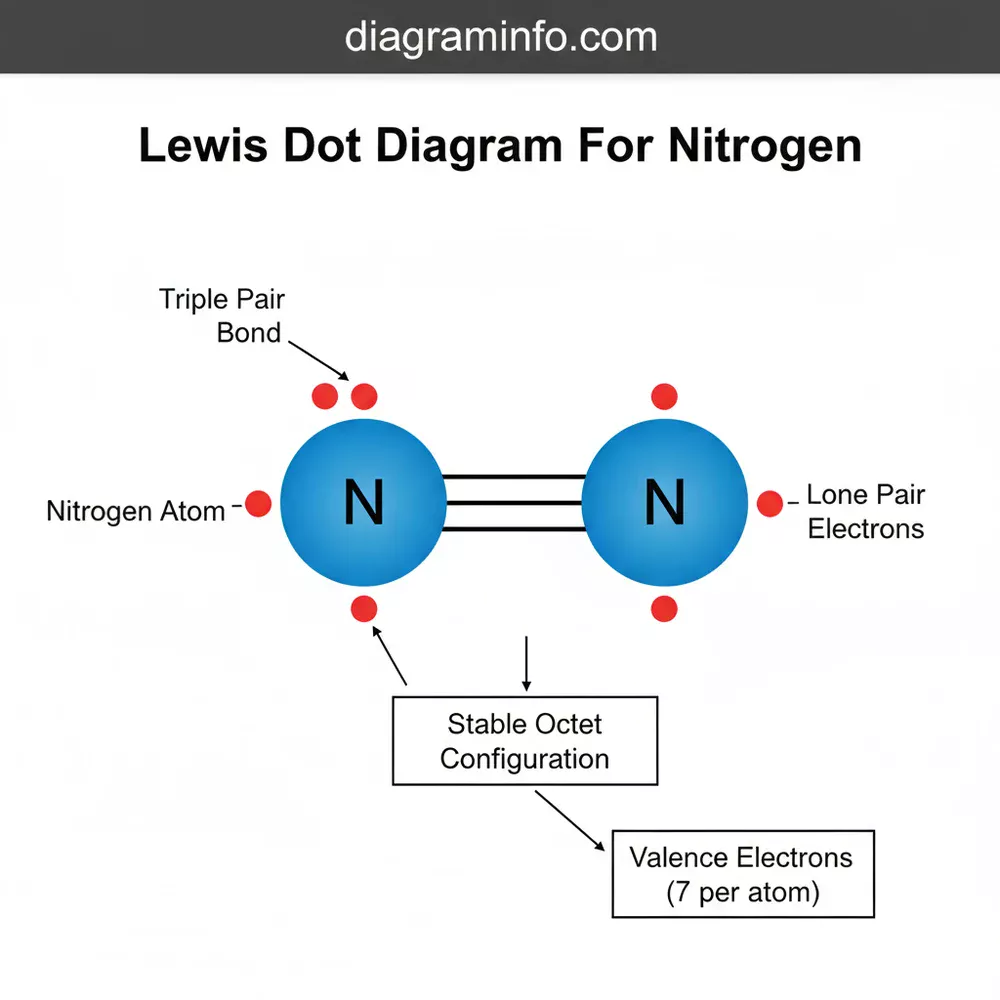
A Lewis dot diagram for nitrogen (N₂) displays two nitrogen atoms sharing three pairs of electrons, forming a triple covalent bond. Each atom also possesses one lone pair. This configuration ensures both nitrogen components achieve a stable octet structure, representing the fundamental molecular layout and electronic system.
📌 Key Takeaways
- Visualize the valence electron arrangement in a nitrogen molecule
- Identify the central triple bond between the two nitrogen atoms
- Note the importance of the lone pairs for atomic stability
- Use this diagram to predict chemical bonding behavior and molecular geometry
- Refer to this layout when balancing chemical equations involving nitrogen gas
Understanding the Lewis dot diagram for nitrogen is a fundamental milestone for students and science enthusiasts alike, as it serves as a gateway to grasping how the most abundant gas in our atmosphere interacts with the world. Having an accurate diagram is not just a classroom requirement; it is a critical tool for predicting chemical reactivity, understanding molecular geometry, and mastering the principles of covalent bonding. This visual overview provides a clear schematic of nitrogen’s valence electrons, offering a blueprint for how this element forms the backbone of DNA, proteins, and fertilizers. In the following sections, you will learn the exact configuration of the nitrogen atom, how to interpret its electronic layout, and the practical steps needed to draw it correctly for both individual atoms and complex molecules.
Nitrogen is located in Group 15 of the periodic table, which immediately tells us that its valence shell configuration contains five electrons. These electrons are the primary components involved in chemical reactions and bonding.
The main layout of a Lewis dot diagram for nitrogen consists of the elemental symbol “N” surrounded by five dots, which represent its valence electrons. To understand this structure, one must first look at the atomic configuration. Nitrogen has an atomic number of 7, meaning it possesses seven protons and seven electrons in its neutral state. These electrons are distributed into two energy levels: the first shell (1s) contains two electrons, while the second, outermost shell (the valence shell) contains the remaining five. The Lewis system focuses exclusively on these five valence electrons because they are the active participants in chemical systems.
When looking at the schematic for a single nitrogen atom, the five dots are arranged in a specific pattern. According to Hund’s rule and the Pauli Exclusion Principle, electrons will occupy empty orbitals before pairing up. In the visual representation, you will typically see one pair of electrons (a lone pair) and three single, unpaired electrons. This configuration is essential because it highlights nitrogen’s “trivalency,” or its tendency to form three covalent bonds to reach a stable octet of eight electrons. The lone pair is often positioned at the top of the symbol, while the three unpaired electrons are distributed on the remaining sides.
In more advanced applications, such as the diatomic nitrogen molecule (N2), the diagram transforms into a more complex blueprint. Here, two nitrogen atoms share three pairs of electrons, creating a triple bond. This is represented by three horizontal lines or six dots placed between the two “N” symbols. Each nitrogen atom retains one lone pair on its outer side. This specific layout explains why nitrogen gas is so stable and unreactive under normal conditions; the triple bond is incredibly strong and requires a significant amount of energy to break. Whether you are looking at a simple atomic overview or a complex molecular schematic, the placement of these dots is the key to unlocking the chemical identity of nitrogen.
Always remember the “6-2 Rule” for diatomic nitrogen: there are 6 shared electrons in the triple bond and 2 lone pairs (one on each atom), totaling 10 valence electrons for the entire N2 system.
To interpret and draw the Lewis dot diagram for nitrogen accurately, follow this detailed step-by-step guide. This process ensures that you account for every component of the atom’s electronic structure and avoid common mistakes associated with valence shell calculations.
-
✓ Step 1: Determine the Total Number of Valence Electrons
The first step in creating any chemical blueprint is identifying the number of valence electrons. For nitrogen, look at its position on the periodic table. As a member of Group 15 (also known as Group VA), nitrogen consistently possesses 5 valence electrons in its neutral state. -
✓ Step 2: Write the Elemental Symbol
Place the letter “N” in the center of your workspace. This symbol represents the nucleus of the nitrogen atom and its inner-shell (core) electrons, which do not participate in bonding. -
✓ Step 3: Distribute the First Four Electrons
Imagine a square box around the “N” symbol. Place one dot on each of the four sides (top, bottom, left, and right). This follows the principle that electrons are negatively charged and repel each other, preferring to remain unpaired as long as empty orbitals are available in the subshell. -
✓ Step 4: Pair the Fifth Electron
Since nitrogen has five valence electrons, you have one remaining after the initial distribution. Place this fifth dot on any side that already has a single dot. This creates one “lone pair” and leaves three “unpaired electrons.” -
✓ Step 5: Verify the Octet Potential
Check your diagram to see how many more electrons are needed to reach the stable number of eight. Nitrogen has three single dots, meaning it needs three more electrons to complete its octet. This confirms why nitrogen typically forms three bonds. -
✓ Step 6: Constructing the N2 Molecule (Optional)
If your goal is to draw nitrogen gas (N2), bring two nitrogen atoms together. Move the three unpaired electrons from each atom into the space between them. These six electrons form the triple bond, while the lone pairs remain on the outside of each nitrogen symbol.
For this task, you will need basic materials: a periodic table for reference, a pencil or digital drawing tool, and an eraser to correct electron placement. No specialized laboratory equipment is required for drawing the diagram, but a firm grasp of atomic numbers is essential.
Do not confuse the total number of electrons (7) with the valence electrons (5). Drawing 7 dots on a Lewis diagram is a common error that will lead to incorrect predictions of molecular geometry and bonding.
Even with a clear guide, users often encounter specific issues when interpreting or drawing the Lewis dot diagram for nitrogen. One of the most frequent problems is the “Octet Obsession,” where students try to force eight dots around a single, isolated nitrogen atom. It is vital to remember that a single nitrogen atom does not have an octet; it only achieves this state by sharing electrons with other atoms. If you see a diagram of a lone nitrogen atom with eight dots, it is likely representing the nitride ion (N3-), which has gained three electrons to become negatively charged.
Another common issue involves the layout of the triple bond in N2. Some beginners draw three separate lines but forget to include the lone pairs. Without the lone pairs, the diagram is incomplete and fails to account for all 10 valence electrons in the system. The diagram helps solve these issues by acting as an accounting system for electrons. If the total number of dots and lines (where each line equals two dots) does not equal the sum of valence electrons from all atoms involved, you know there is a mistake in your configuration. If you find yourself unable to balance the electrons in more complex nitrogen-containing molecules like nitrate (NO3-) or ammonium (NH4+), it may be time to seek help from a chemistry tutor or a professional academic resource, as these involve resonance structures and formal charges.
To excel in chemical modeling, consider these best practices for working with nitrogen diagrams. First, always calculate the formal charge for each atom in your schematic. For nitrogen, the formula is: (Valence Electrons) – (Non-bonding electrons) – (1/2 Bonding electrons). In a neutral nitrogen atom in ammonia (NH3), this would be 5 – 2 – 3 = 0, indicating a stable structure. This mathematical check ensures your visual layout is physically accurate.
In terms of maintenance of your chemistry knowledge, regularly practice drawing nitrogen in various hybridized states. Nitrogen can exist in sp3 hybridization (as in ammonia), sp2 (as in pyridine), or sp (as in hydrogen cyanide). Understanding how the Lewis dot diagram for nitrogen adapts to these different configurations will significantly improve your ability to visualize three-dimensional molecular geometry. For those looking to save time during study sessions, utilizing digital molecular modeling kits can provide high-quality, three-dimensional views of these electron clouds that a 2D diagram cannot fully capture.
When selecting components for your study—such as textbooks or software—opt for those that emphasize the VSEPR (Valence Shell Electron Pair Repulsion) theory. This theory builds directly upon the Lewis diagram by explaining how the lone pair on the nitrogen atom pushes the bonding pairs away, resulting in the characteristic pyramidal shape of molecules like ammonia.
The Lewis dot diagram for nitrogen is more than just a cluster of dots and letters; it is a sophisticated overview of the element’s potential to create life-sustaining molecules. By identifying the five valence electrons and understanding their specific layout, you gain the ability to predict the behavior of nitrogen in a wide array of chemical systems. From the simple atomic schematic to the robust triple-bonded blueprint of N2, mastering this diagram is essential for any serious student of the sciences. By following the step-by-step guides and avoiding common pitfalls like incorrect electron counts, you can ensure your chemical models are both accurate and informative. Whether you are troubleshooting a complex bonding issue or simply starting your journey into atomic structure, the nitrogen Lewis diagram remains one of the most reliable and powerful tools in your scientific toolkit.
Frequently Asked Questions
Where are the lone pairs located?
In the nitrogen molecule, the lone pairs are located on the outer side of each nitrogen atom, opposite the triple bond. This specific layout balances the electronic system. Correctly identifying this component is essential for predicting how the nitrogen molecule will interact with other chemical substances during reactions.
What does this Lewis dot diagram show?
The Lewis dot diagram for nitrogen shows the arrangement of valence electrons around the atomic nuclei. It illustrates the triple covalent bond formed between two nitrogen atoms and the lone pairs that complete the octet. This visualization helps in understanding the chemical stability and bonding configuration of nitrogen.
How many valence electrons does nitrogen have?
A single nitrogen atom has five valence electrons. In the N2 molecule, two nitrogen atoms provide a total of ten valence electrons. These electrons are distributed to form three shared pairs (a triple bond) and two unshared pairs (lone pairs) within the molecular system and structure.
What are common errors in drawing this diagram?
A common error in drawing this diagram is failing to provide a triple bond or forgetting the lone pairs. If the total electron count does not equal ten, the configuration is incorrect. This mistake often leads to an incomplete octet, misrepresenting the stability of the nitrogen component.
Can I draw this diagram myself?
Yes, you can easily draw this diagram by following the octet rule. Start by counting the total valence electrons available in the nitrogen system. By placing the atoms and distributing the electrons into bonds and lone pairs, you can create an accurate layout of the nitrogen molecule’s structure.
What tools do I need for this task?
To draw a Lewis dot diagram for nitrogen, you simply need a periodic table, a piece of paper, and a pencil. The periodic table helps you identify the number of valence electrons, while the drawing tools allow you to sketch the molecular layout and electron configuration clearly.